Design. Validate.
Record. Execute.
Digitize your lab operations with Logilab ELN, a cloud-based electronic lab notebook (ELN) software accessible through any browser. Combine the simplicity of spreadsheets with advanced AI assisted scientific documentation, multi-step protocol execution, and integrated sample management.
Achieve up to 35% faster documentation, 50% improved audit readiness, and reduce transcription errors by 30% — all while staying compliant with 21 CFR Part 11 and Annex 11
faster collaboration across QC and R&D teams
fewer transcription errors
reduction in documentation time
improvement in audit preparation speed
Why Modern Labs choose Logilab ELN
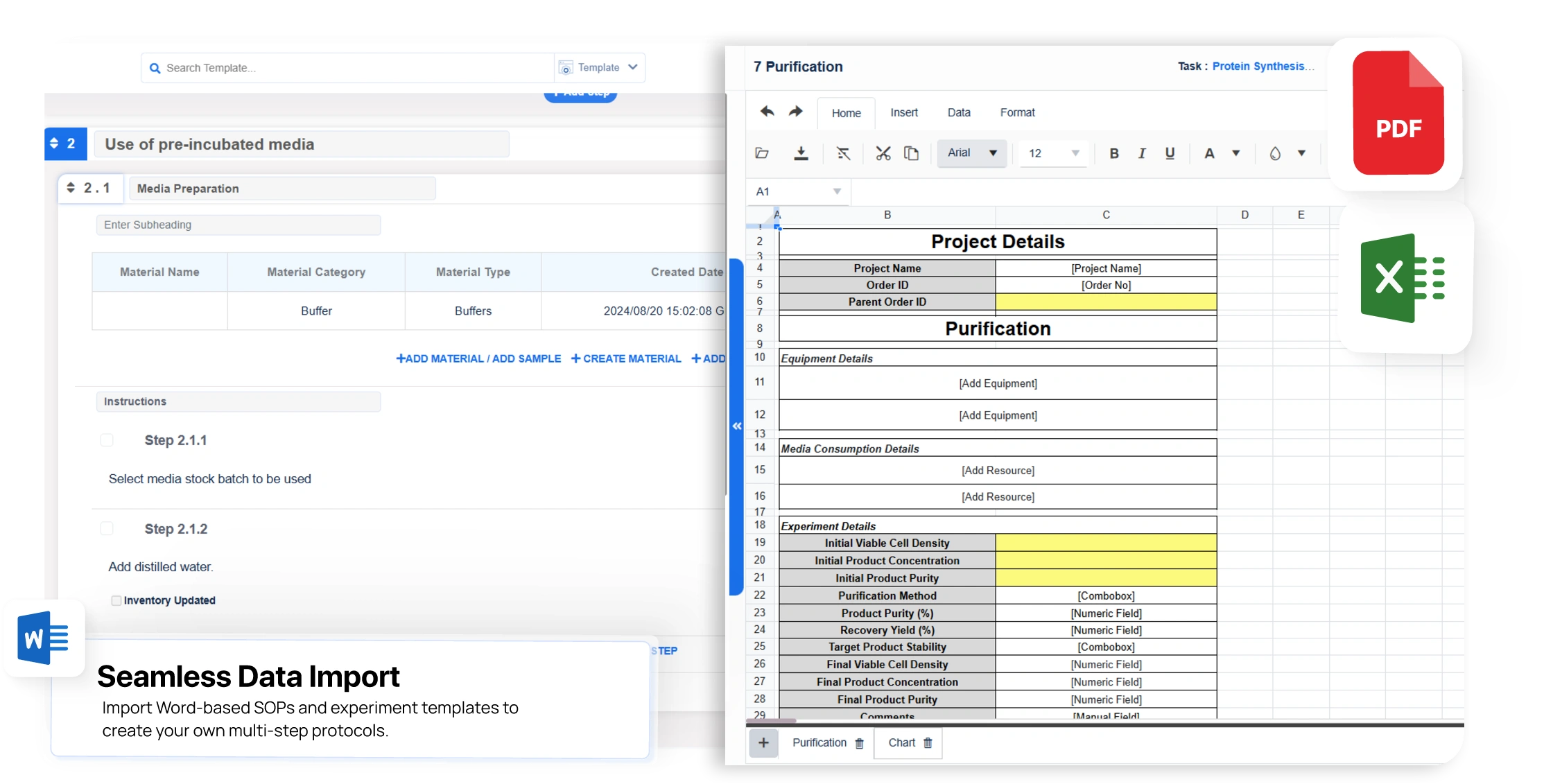
Flexible Labsheet & Multi-Step Protocol Design
- Create customized labsheet templates with a familiar spreadsheet-style interface inside the ELN
- Build multi-step protocols to standardize complex workflows across scientific documentation and experimental procedures
- Maintain version control for both labsheets and protocols to preserve data integrity and traceability
- AI-powered template creation from legacy files to help optimize data migration and reduce repetitive design tasks
Seamless Experiment Execution
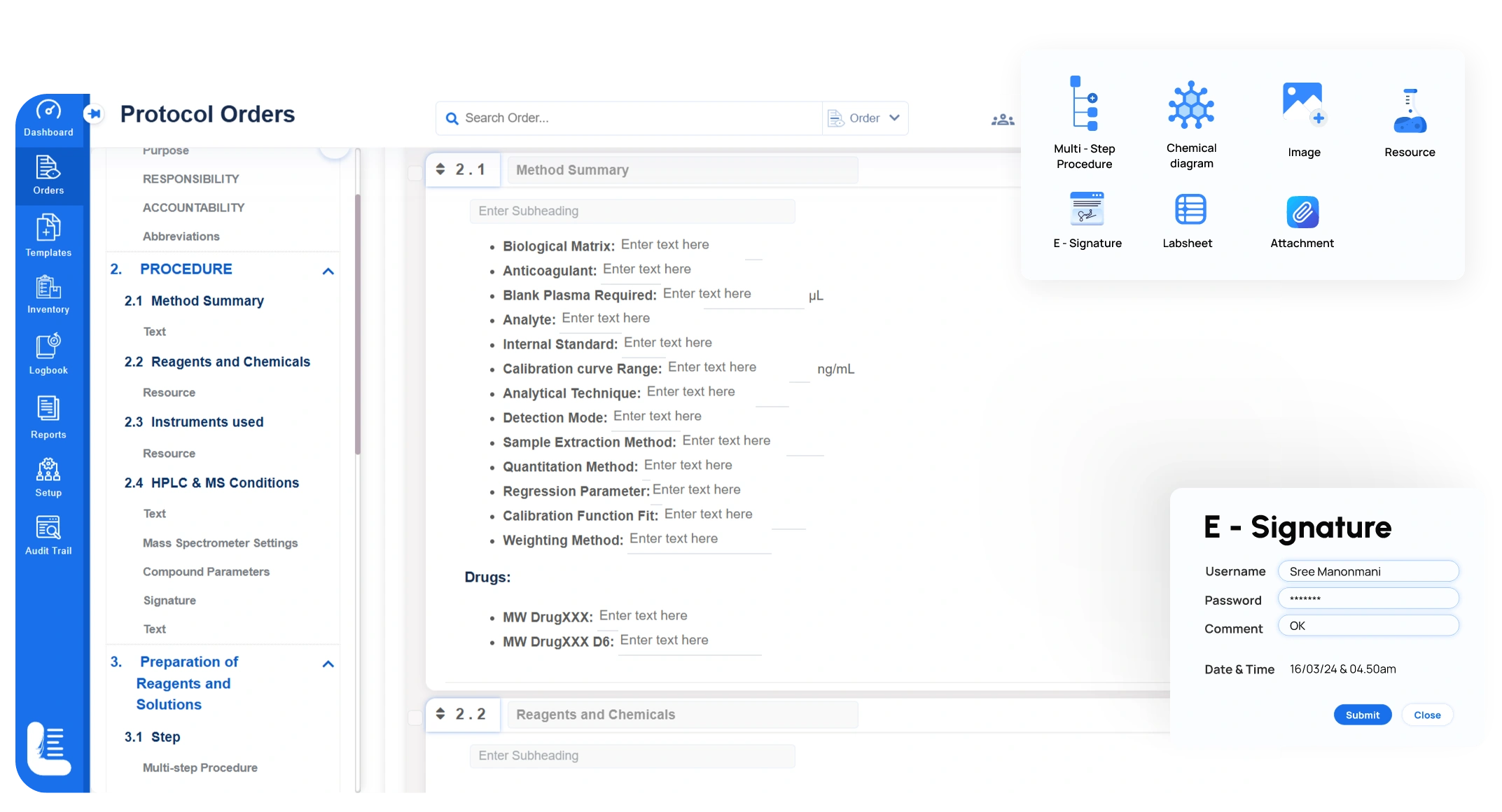
- Execute experiments using labsheets or multi-step digital workflows directly in the ELN
- Capture experimental data in real time, linking results to samples, instruments, and operators
- Apply electronic signatures and follow multi-stage approval workflows for GxP compliance
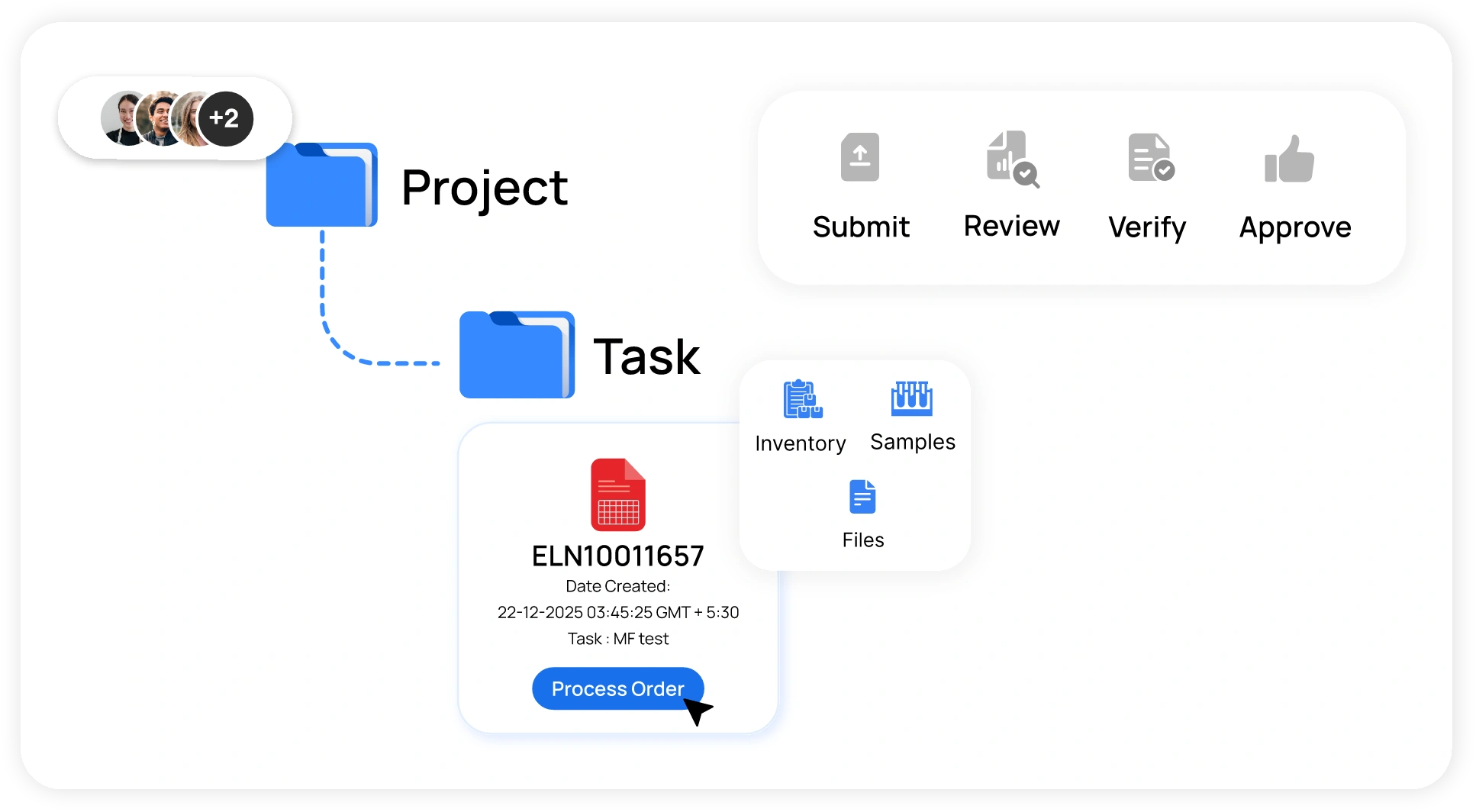
Project Management for R&D Labs
- Plan, assign, and monitor lab activities across projects directly in the cloud ELN
- Link experiments, samples, and results to specific project milestones
- Track progress, timelines, and responsibilities for better cross-team coordination
- Centralized project dashboards give QC and R&D teams real-time visibility on experiment status and pending approvals
Discover, Document & Execute any Lab task

Design Custom
Labsheets & Protocols
Run Experiments &
Projects


Execute Tasks
Validate Results


Design Custom Labsheets & Protocols

Run Experiments and Projects

Execute Tasks

Validate Results
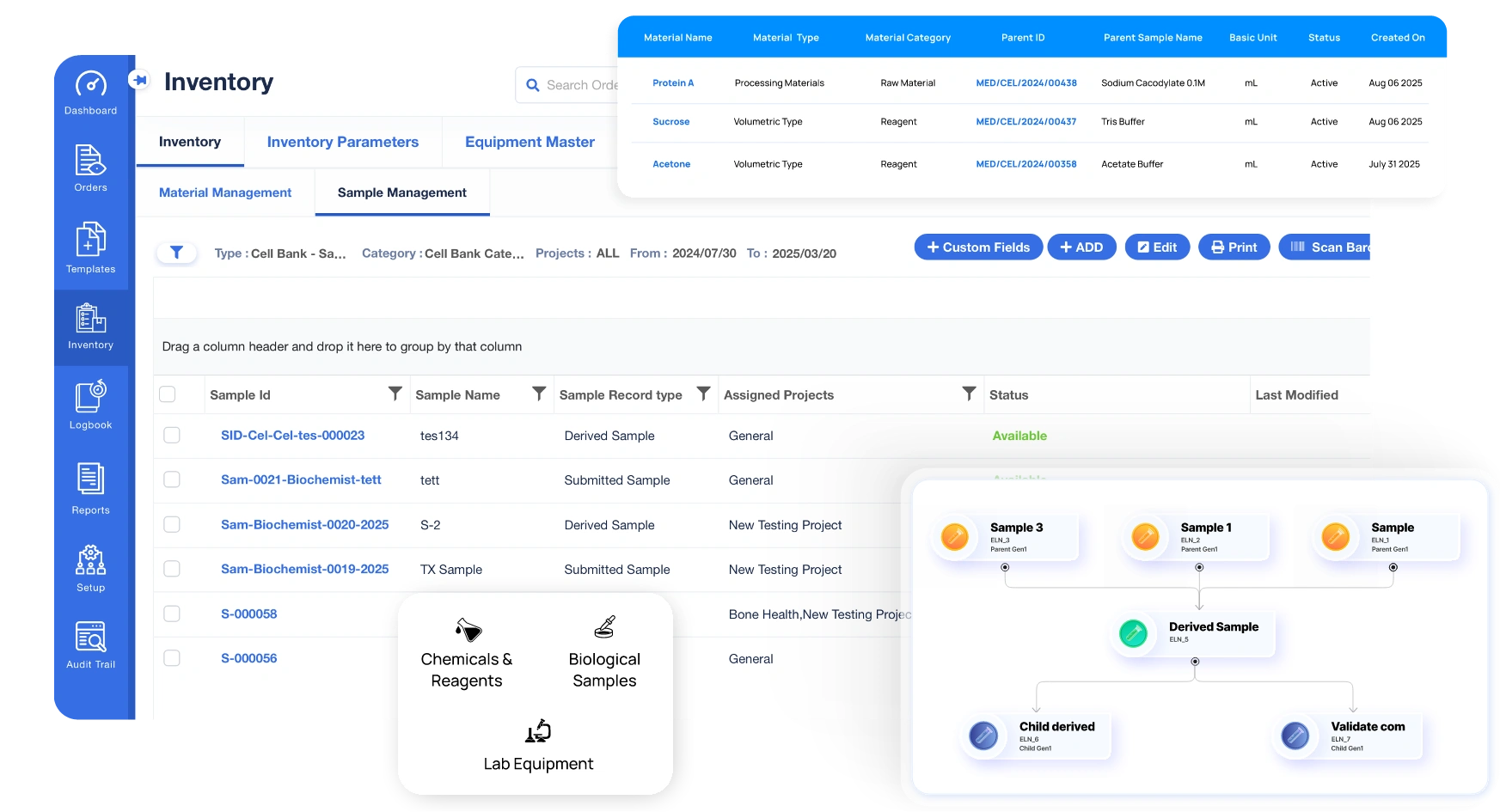
Integrated Sample & Material Management
- Create & manage materials samples in the ELN for research, QC, and clinical workflows
- Track sample usage and hierarchies - maintain end-to-end traceability from registration to disposal
- Track chemicals, reagents, and consumables with barcode labelling
- Record usage history and define your own metadata fields for every sample
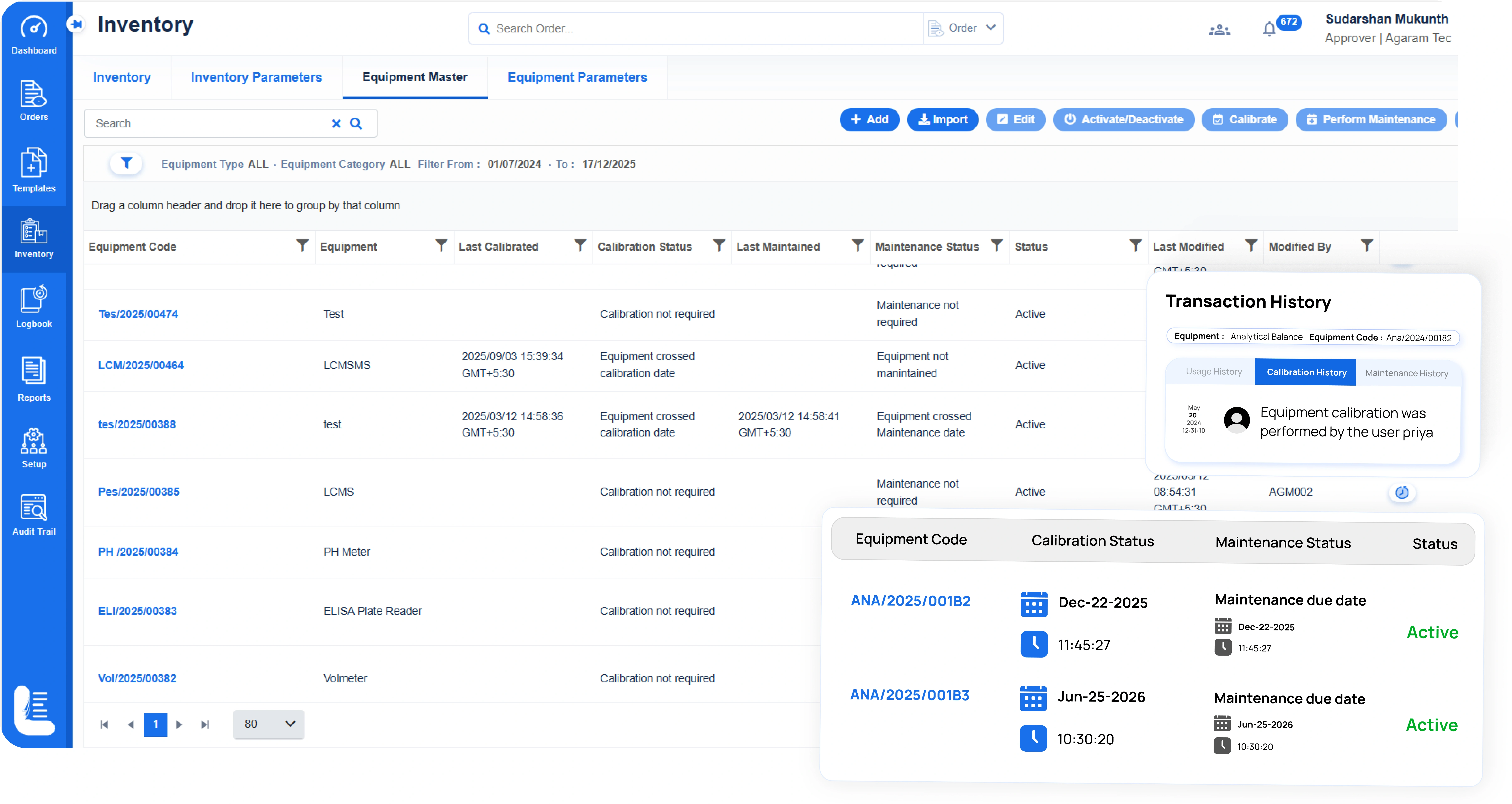
Built-in Equipment Management
- Track Instrument usage across experiments
- Manage instrument calibration, maintenance, and qualification intervals & records
- Ensure calibrated and maintained instruments are used in experiments for compliance-ready workflows.
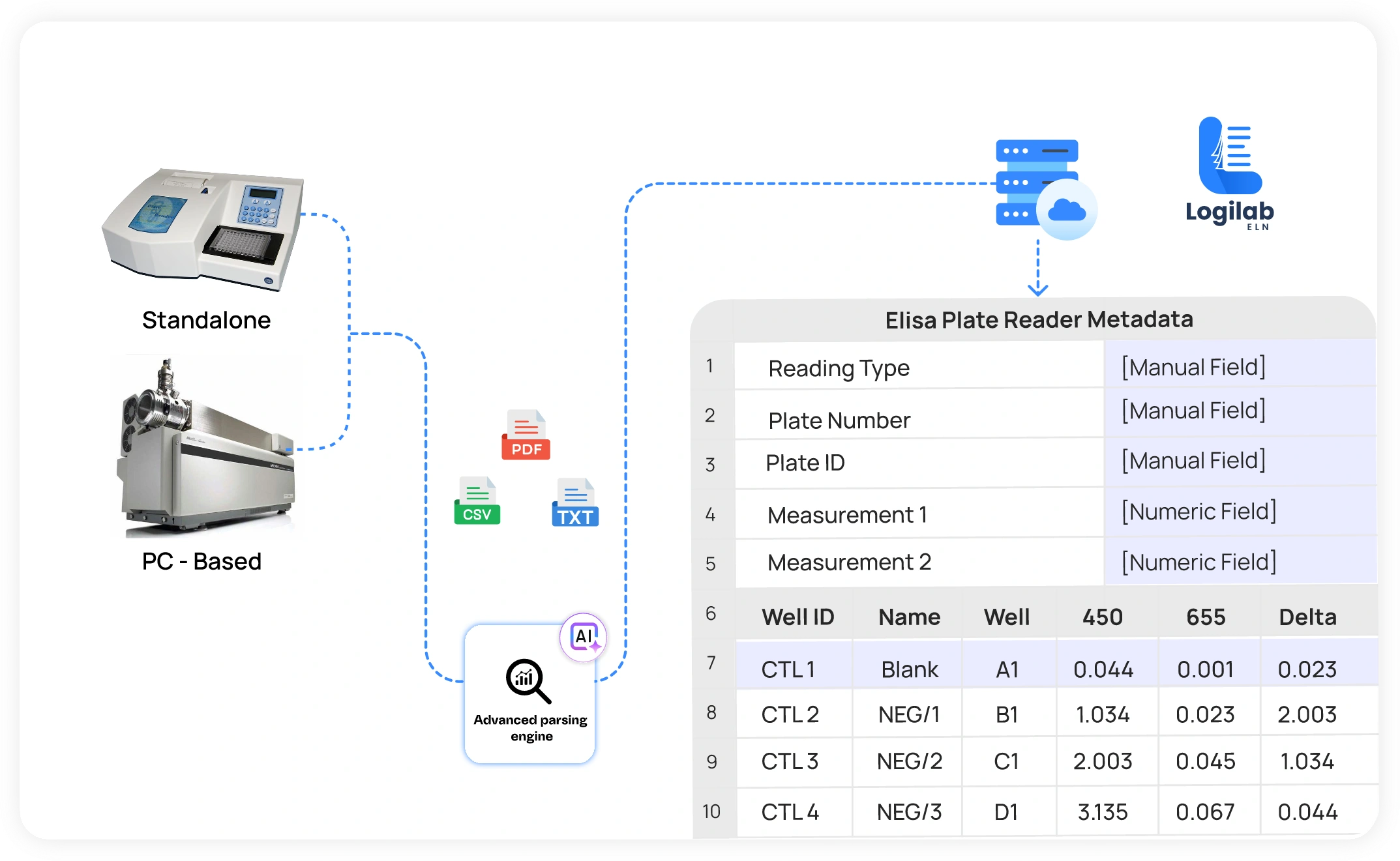
Connect Instruments to your Electronic Lab Notebook
- Support for RS232, TCP/IP, and cloud-based instrument data flows to eliminate manual transcription
- Link instrument readings directly to experiments and samples in your ELN for full traceability.
- Manage instrument qualification, calibration, and preventive maintenance schedules — ensuring only compliant instruments are used in active experiments
- AI-based parsing engine with Logilab SDMS to extract data accurately & easily from instruments
Compliance
You Can
Trust
supports
ALCOA+ Data Integrity Principles
for reliable, audit-ready data
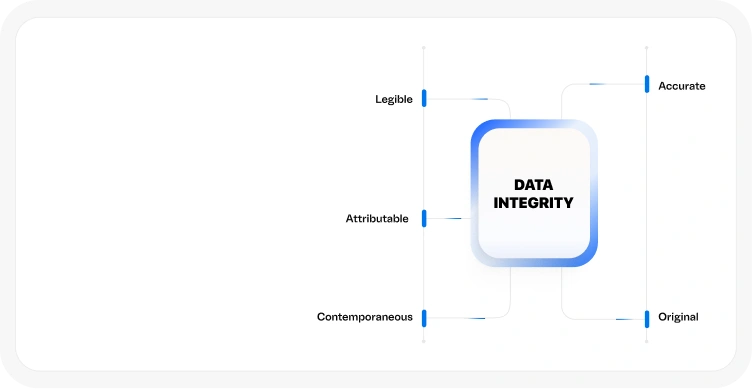
Fully aligned with 21 CFR Part 11, EudraLex Annex 11, and GLP guidelines

Features include audit trails, e-signatures, and role-based access controls

supports
ALCOA+ Data Integrity Principles
for reliable, audit-ready data

Fully aligned with 21 CFR Part 11, EudraLex Annex 11, and GLP guidelines
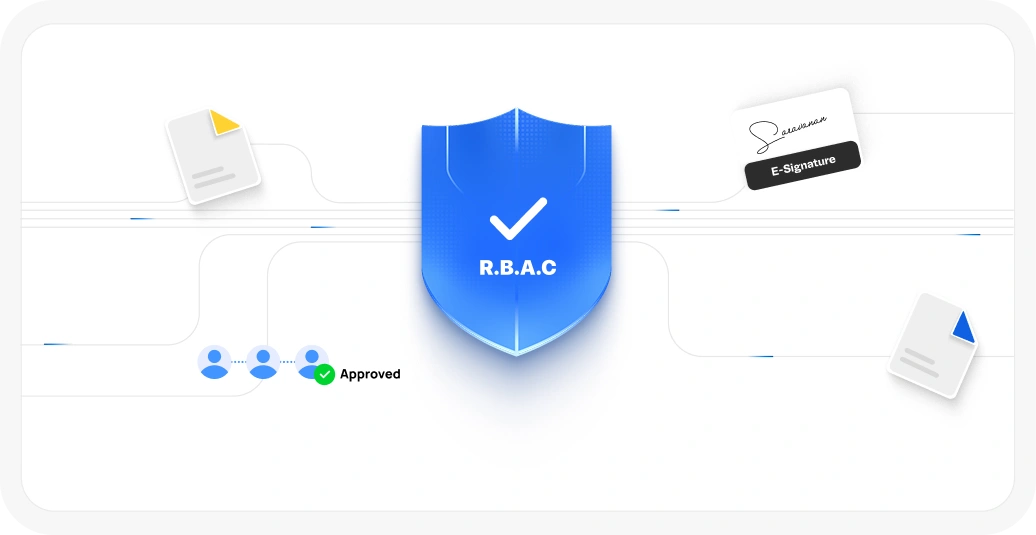
Features include audit trails, e-signatures, and role-based access control
A Connected Laboratory Ecosystem

- Out-of-the-box integration with Logilab SDMS for automated instrument data capture
- Seamless connectivity with Qualis LIMS for unified sample and data management
- API support for integrating with third-party ELN, LIMS, and scientific data systems
- AI-ready architecture ensures future compatibility with emerging machine learning and automation tools.
Key Benefits at a Glance
Paperless Lab Operations
Eliminate manual transcription errors
Multi-Format Support
Labsheets & multi-step protocols for flexible experiment design and easy import of legacy data
Regulatory Compliance
21 CFR Part 11, Annex 11, GLP ready
Enhanced Collaboration
Multi-user access, real-time updates
Sample Management
Centralized registration, tracking, and usage history
Data Integrity
ALCOA+ principles, audit trails, secure access
Customer Success Snapshot

We delivered the first major milestone of our e-Lab Vision project by launching Logilab ELN across our Biologics and MMIC Technical Teams, replacing legacy systems and moving to a secure, cloud-based platform
Suzanne Buglass – Project Manager ( Source )

We delivered the first major milestone of our e-Lab Vision project by launching Logilab ELN across our Biologics and MMIC Technical Teams, replacing legacy systems and moving to a secure, cloud-based platform
Suzanne Buglass – Project Manager
Get Started with Logilab ELN Free Edition
With the Free Edition, you can try essential features like labsheet & multi-step protocol creation, sample tracking, & secure cloud — all designed to boost productivity and ensure compliance
Frequently Asked Questions
Digitize . Simplify . Organize
Kickstart your paperless lab with Logilab ELN



















