
Key roles of ELN in Laboratories
An electronic laboratory notebook (ELN) is a computerised system that includes tools for creating and storing experimental protocols, results, and research findings on a digital platform. ELNs have been developed

An electronic laboratory notebook (ELN) is a computerised system that includes tools for creating and storing experimental protocols, results, and research findings on a digital platform. ELNs have been developed

If you’re on the fence about what exactly the Logilab Scientific Data Management system can do for your research laboratory, stick around as we deep dive into its powerful instrument
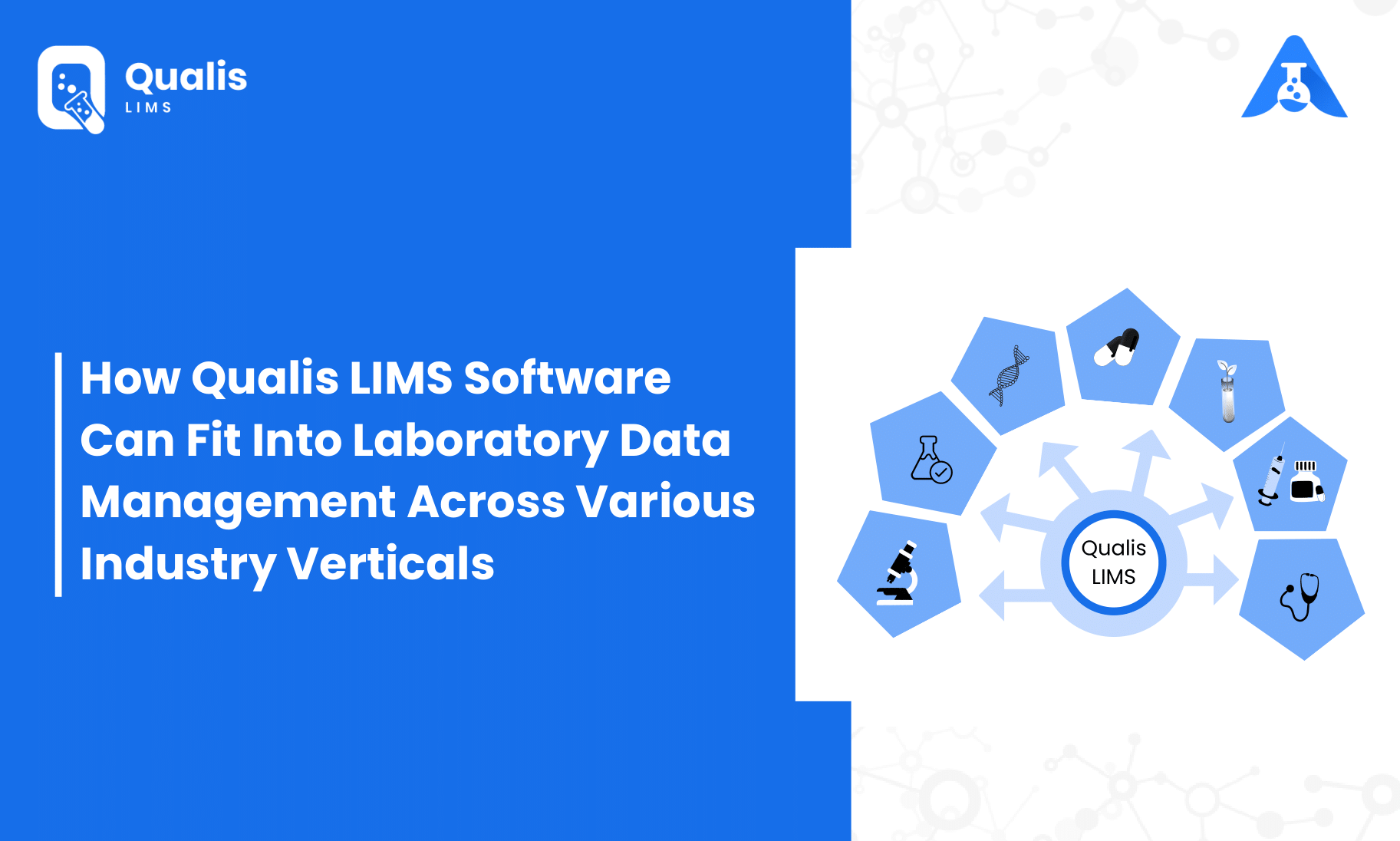
In a typical laboratory, the amount of data generated grows over time, increasing storage and processing constraints exponentially. Once the complexity gets to a certain threshold, it becomes a nearly

What do you think is the role of a laboratory information management system (LIMS) in a modern laboratory? Are you considering using a LIMS for your laboratory? Here is some

If you have been a LIMS vendor or are a company that falls under the life sciences field for quite a while now, you probably already know that regulatory compliance
The importance of data traceability and accuracy couldn’t be underscored more as far as any kind of research is concerned. It serves as the primary way for studies to be

Every modern laboratory in the industry is constantly focusing to enhance its operational efficiency, decrease costs, control regulatory compliance, and improving quality. Apart from this, the rise of Contract Research

Before getting into the steps involved in finding the perfect SDMS, it is essential to understand the meaning of SDMS. A scientific data management system (SDMS) acts as a centralized document
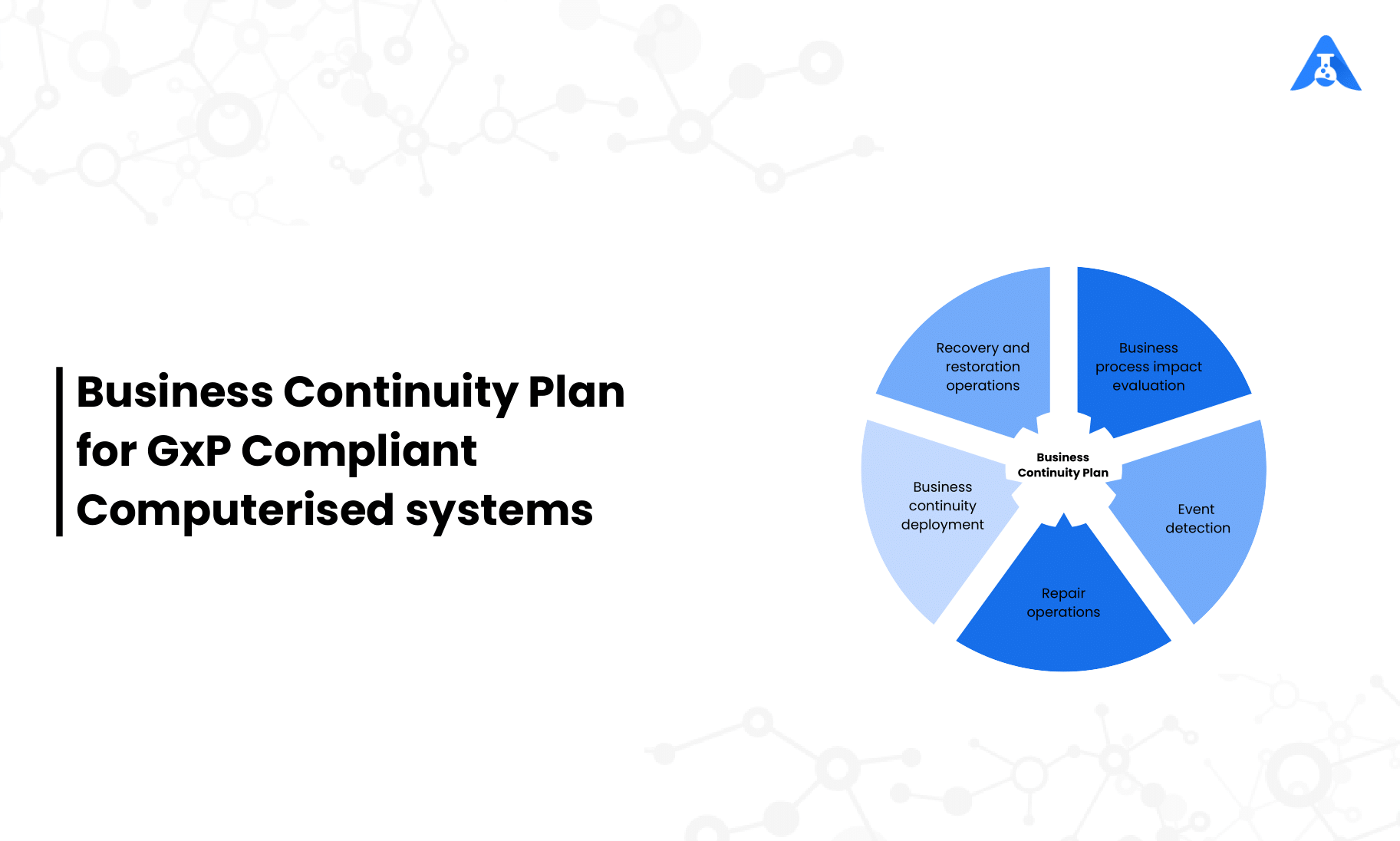
For the availability of computerised systems critical processes, provisions should be made to ensure continuity of support for those processes in the event of a system breakdown (e.g., a manual

Data Integrity Data integrity, an area of increasing priority in today’s laboratory world and “The focus area” when it comes to regulatory compliances such as 21 CFR Part 11 and