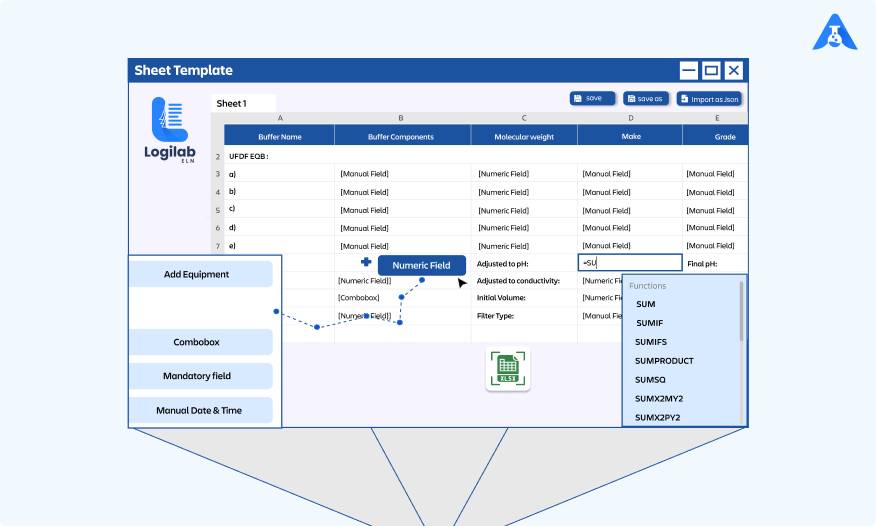
Centre for Process Innovation (CPI), UK, Adopts Agaram’s Logilab ELN to Drive R&D Innovation
Agaram Technologies is pleased to announce that the Centre for Process Innovation (CPI), a UK-based technology innovation center, has chosen Logilab ELN as its preferred Electronic Lab Notebook solution. The